Showing results for Anna Carrera
-
![tyler-trent-home tyler-trent-home]()
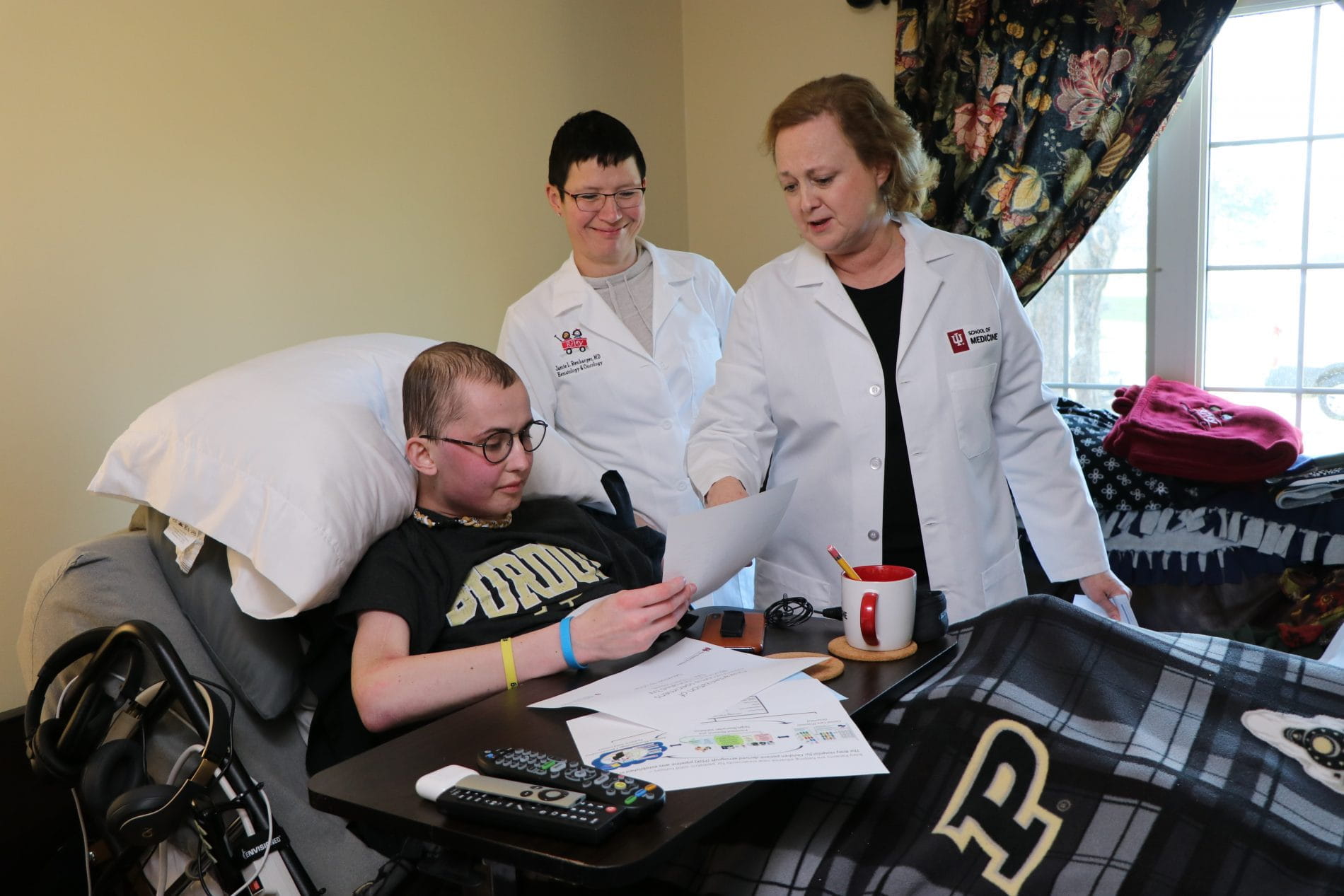
Pediatric researchers receive two new grants to support work based on Tyler Trent's tumor cells
Two new grants supporting osteosarcoma research have been funded at IU School of Medicine, building on work that has been previously established based on Tyler ...
Anna Carrera | Nov 28, 2022 -
![Young cancer survivors]()
Researchers secure grant to better understand barriers to cancer survivorship for adolescents and young adults
A team of researchers at IU School of Medicine have received $100,000 in pilot funding to research how best to deliver cancer survivorship healthcare to ...
Anna Carrera | Nov 03, 2022 -
![7418-John, Chandy 7418-John, Chandy]()
Researchers study effects of malaria and preventative treatments in young children
Researchers are learning more about how children’s brain development may be affected when their mother has malaria while she is pregnant, as well as the ...
Anna Carrera | Oct 25, 2022 -
![]()
New director named for Office of Clinical Research
Carmel Egan, PhD, has been named the director for the Office of Clinical Research (OCR). As the new director, Egan will provide leadership and direction ...
Anna Carrera | Sep 22, 2022 -
![research reception]()
Researchers recognized for receiving federal grants in 2021
IU School of Medicine researchers who received federal grants in 2021 were recognized during a special event on Thursday, April 21 at the VanNuys Medical ...
Anna Carrera | Jun 16, 2022 -
IU School of Medicine and Lilly partnership achieves success in science and oversight
In 2020, a partnership between Indiana University School of Medicine and Eli Lilly and Company was created to collaborate in specific areas of scientific development. ...
Anna Carrera | Apr 13, 2022 -
![13390-Lahiri, Debomoy 13390-Lahiri, Debomoy]()
New findings could contribute to the early detection of Alzheimer's disease
IU School of Medicine distinguished professor, Debomoy K. Lahiri, PhD, and his team found that “mis”-production of amyloid precursor protein (APP) in the brain contributes ...
Anna Carrera | Apr 12, 2022 -
![child vaccine]()
Pediatric researchers urge families to vaccinate kids against COVID-19 now
Three pediatric researchers, including one from IU School of Medicine, are encouraging parents to get their children vaccinated against COVID-19 as soon as possible. The ...
Anna Carrera | Mar 29, 2022 -
![Multiple myeloma roundtable with Rafat Abonour and Dorothy Frapwell]()
Third IU Precision Health Initiative webinar focuses on multiple myeloma progress
The third IU Grand Challenges webinar about the Precision Health Initiative (PHI) took place in October, focusing on progress in studying and treating multiple myeloma, ...
Anna Carrera | Nov 11, 2021 -
![James Wood]()
New publication means improved outcomes for children with Kawasaki disease
A new publication with contributions from an IU School of Medicine researcher could change the way doctors treat children diagnosed with Kawasaki disease. James Wood, ...
Anna Carrera | Nov 09, 2021
Anna Carrera
Anna Carrera is the research communications manager for Indiana University's Precision Health Initiative, IU School of Medicine and the Indiana Clinical and Translational Sciences Institute. She joined the team in June 2019 after working as a TV news rep...