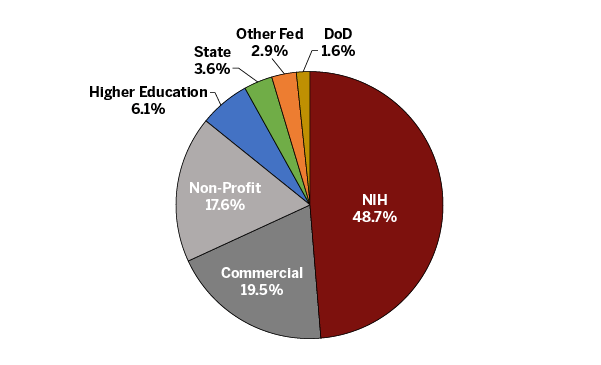
IU School of Medicine Researchers have secured an impressive $450 million in external grant funding over the last 12 months through a variety of funding sources. The pie chart below shows the percentage of funding attributed to each funding mechanism. This high-level breakdown illustrates a diverse portfolio of grant funding that supports the wide breadth and depth of research conducted at the school.
The graphic also shows that nearly half of the school’s $219 million in research funding is from the National Institutes of Health (NIH). All external funding that supports our research mission is important to our success; however, NIH funding is a key metric used nationally to evaluate and compare academic institutions. An enterprise goal of IU School of Medicine and IU Health is for the school to be among the top 25 medical schools in the country for NIH funding.
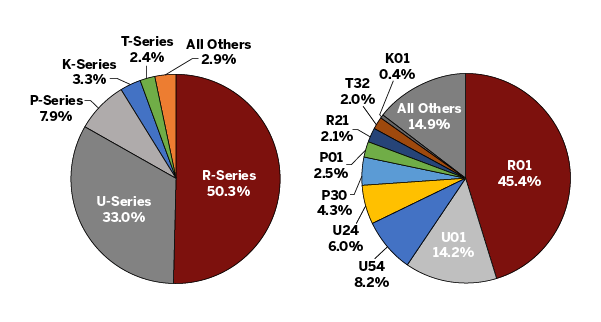
The charts below provide a more detailed view of the NIH grants funded in the school. In the pie chart on the left, the NIH grants shown in red above are broken out to illustrate the different types of NIH grants. The pie chart on the right provides further granularity through the depiction of the most frequently awarded grant mechanisms. For example, among the R-series (Research) grants, the majority of our school funding is from R01 grants. The average R01 award has been steadily increasing and the proportion of the limited modular R01s ($250K direct costs/year) has been decreasing. Cooperative grants (U grants) provide one-third of our NIH funding. P grants consist of program project grants and center grants. These grants include groups of investigators and support important cores on campus. Trainees at various levels are supported through training (T series) and career development (K series) grants.
Receiving your first research grant (R01) from the NIH as a principal investigator is a major achievement. I want to recognize (below) our IU School of Medicine colleagues who were awarded their first R01s between January 1, 2021 and July 1, 2021. Please congratulate each of them! I look forward to hearing more about their new research discoveries.

As a reminder, there is one more month until mandatory data management plans are due. We are seeing progress in submissions, but we are not at 100% yet. The forms are award-based and due no later than August 31, 2021. Assistance with the requirement is available.

Tatiana Foroud, PhD
Executive Associate Dean for Research Affairs,
Indiana University School of Medicine
Executive Vice President of Academic Affairs for Clinical Research, IU Health
