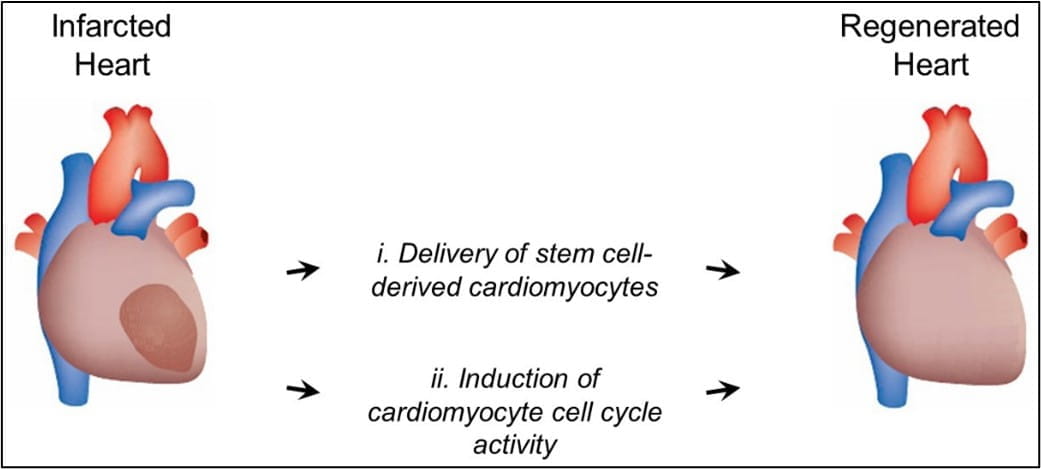
Acute Myocardial Infarction (MI) is a leading cause of death worldwide. Survivors of MI usually develop scar tissue in the infarcted regions of the heart, which leads to inefficient cardiac function and other associated pathology. Such patients could benefit greatly from regenerative medicine, which aims to replace the scar with healthy muscle cells, thus restoring normal function. The Myocardial Regenerative Biology Research Program (MRBRP) at KCVRC is investigating novel ways to repair damaged heart tissue utilizing stem cell or cell-free therapies.
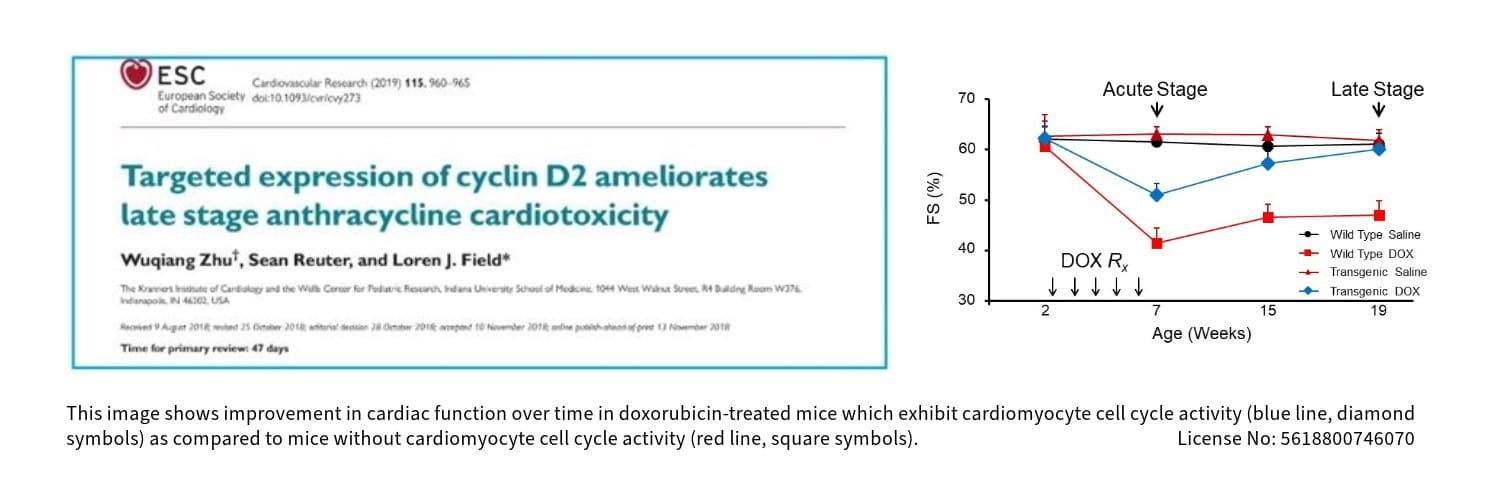
This program, led by molecular biologist Loren J. Field, PhD, has a long-standing track record in developing genetic models and strategies to monitor the intrinsic rates of cardiomyocyte cell cycle renewal in normal and injured adult hearts, as well as developing strategies to induce regenerative growth following myocardial injury.